
8 The covalent character of hydrogen bonding also has been described by Coulson as “covalent-ionic resonance” 9 and by several others, as well as from a valence bond perspective, as “three-center-four-electron” interactions. A hydrogen bond X-H…Y may be viewed as a donor–acceptor orbital interaction, in which a set of lone pairs on Y donate into the antibonding orbital of X-H. 7 These early depictions of hydrogen bonds were developed from Lewis’ theory 6 for valence and bonding and hinted at the covalent character (i.e., orbital interaction) of hydrogen bonds. They recognized that ammonium hydroxide ( Figure 2b) is another example in which the “ union is fairly strong,” explaining that “… the hydrogen nucleus held between two octets constitutes a weak bond.” Huggins proposed the term “ hydrogen bridges” to describe the sharing of an H atom between two molecules. They noted that ammonia readily adds a hydrogen, hydrogen chloride readily loses one, but water could add or lose a hydrogen, and therefore a hydrogen could be shared between two water molecules and bind two molecules together ( Figure 2a). 
In 1920, Latimer and Rodebush 1 (along with the unpublished works of Huggins a year earlier, Box 1) first related the idea of electronegativity and bond polarity to the sharing of a hydrogen atom between two atoms. 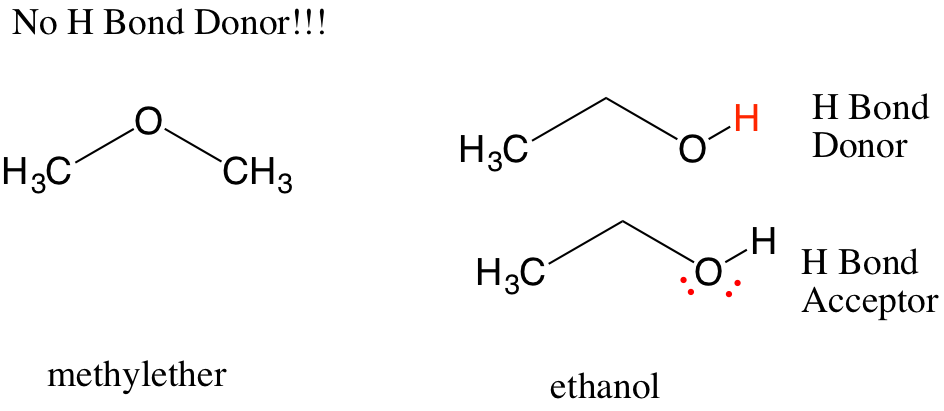
Discussion focuses on how molecular structure affects hydrogen bond strength, and on showcasing selected applications of hydrogen bond design principles. Debates touched on but not discussed in detail include the physical origins of hydrogen bonds and the physical explanations of each design principle. This review presents an overview of hydrogen bond design principles, based on five structural features: electronegativity effects (Section 2), steric effects (Section 3), electrostatic effects (Section 4), π-conjugation effects (Section 5), and cooperativity effects (Section 6). These events happened at the height of early applications of crystallography in chemistry and set the stage for the discovery of many hydrogen bond design principles (1990’s–2000’s), which are now routinely applied to areas of recognition, catalysis, and assembly in organic and supramolecular chemistry. Recognizing that a hydrogen nucleus shared between two atoms could largely influence the three dimensional structure of molecules and molecular fragments initiated a contentious race among multiple groups toward unveiling the double helix DNA structure (1953, Watson–Crick), 4 the α-helix structure (1951, Pauling–Corey–Branson) 5 and the β-sheet structure (1950, Pauling–Corey) 6 of proteins, along with surveys and developments of hydrogen bonding patterns in organic crystals (1950’s–1960’s). Lewis’ theory 3 of valence and chemical structures (1916)-so that the idea of a hydrogen bond could be conceived, and (b) the discovery of X-ray (1895)-so that a hydrogen bond, that is, close contact between proton-sharing atoms, could be observed. Leading up to the magnum opus of Latimer and Rodebush’s proposal of hydrogen bonding, two events in the early 1900’s steered the direction of hydrogen bond research during the first half of the 20th century: (a) G. Since the 1920 report of Latimer and Rodebush, 1 regularity in hydrogen bonding patterns were recognized, hydrogen bond design principles were developed, and it became possible to explain and imagine the structures and functions of many hydrogen bonding systems. 2 From the varying strengths and the directionality of hydrogen bonds, emerges the opportunity for chemical design. They can be as weak as less than a kilocalorie per mole, they can be as strong as half the association of a single C-C bond (e.g., the − interaction is about 40 kcal/mol), and the directionality of hydrogen bonds gives a clue to how molecules and molecular fragments might arrange in space. Hydrogen bonding interactions stand at the crossroad between weak noncovalent bonding and strong covalent bonding. Such an explanation amounts to saying that the hydrogen nucleus held between two octets constitutes a weak ‘bond’.” (Latimer WM and Rodebush WH, 1920) 1. a free pair of electrons on one water molecule might be able to exert sufficient force on a hydrogen held by a pair of electrons on another water molecule to bind the two molecules together. “Water…shows tendencies both to add and give up hydrogen, which are nearly balanced.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
